|
Watch our recent webinar to learn more about important considerations for keeping eCOA off the critical path for clinical trial startups.This Notice encourages investigators with expertise and insights into this area of Alcohol Research to begin to consider applying for this new RFA. Risk assessments around COA use can begin at the protocol design stage, and with a centralised management approach, can be mitigated to keep eCOA off the critical path. Therefore, early planning of an eCOA implementation strategy can significantly help stay on track with critical study milestones. In study startup timelines, every day matters. Ensure everyone is always on the same pageĮverything happening in the industry and in the world suggests that electronic data collection is here to stay and will play an increasingly important role in clinical trials and drug development.Discuss changes, create solutions and communicate frequently.Regular weekly meetings with the service providers and preferably study startup and clinical leads to review the eCOA implementation plan status.Create an eCOA project team with members across the stakeholder groups and apply an agile-like methodology to manage project plan implementation. All the parties involved need to be on the same page before moving forward.īe prepared that everything discussed so far will likely change significantly, tasks will take longer than expected and study startup targets will shift. For example, the screenshots may not be ready in time for submission, discuss the option of submitting paper-version followed by screenshots, etc. The plan will highlight at an early stage any downstream risks in eCOA implementation which can be discussed and mitigated at the project outset. All the stakeholders need to collectively review the plan. The project plan coordinates each service provider’s milestones in alignment with the Sponsor’s study timelines. Document any risk and assumptions in relation to the above.Understand each service provider’s delivery timelines.Define study startup (project) targets (e.g., planned submission dates and site activation dates).To create a successful project plan, the project manager needs to involve all the stakeholders and gather all the data that will impact the delivery of that project early in the process. The goal is to create a single project plan around the Sponsor’s study milestones led by one eCOA project manager responsible for coordinating the different services and parties. What are the keys to a successful eCOA implementation? For example, if patient-facing eCOA material is not ready in time, it may cause delays to ethics submissions, and the first patient can only be enrolled at the site if the eCOA system is ready to collect the data. Without coordination, trial startup milestones may be delayed. 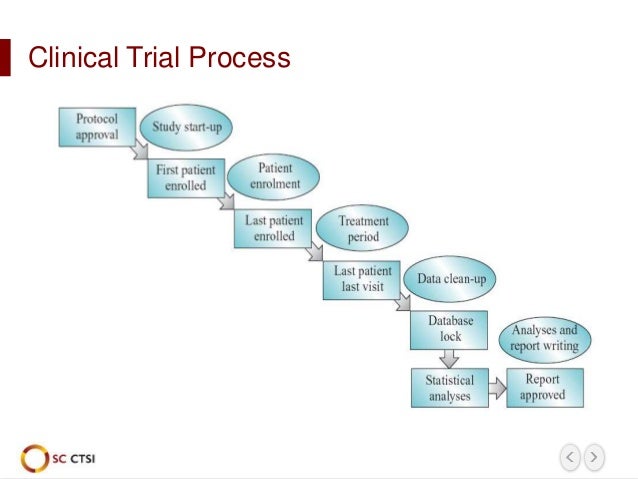
The key risk in eCOA implementation is the coordination of the deliverables for these service areas. 
Most likely, the approval from a copyright holder at different stages of the development process of the electronic versions of the COAs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
